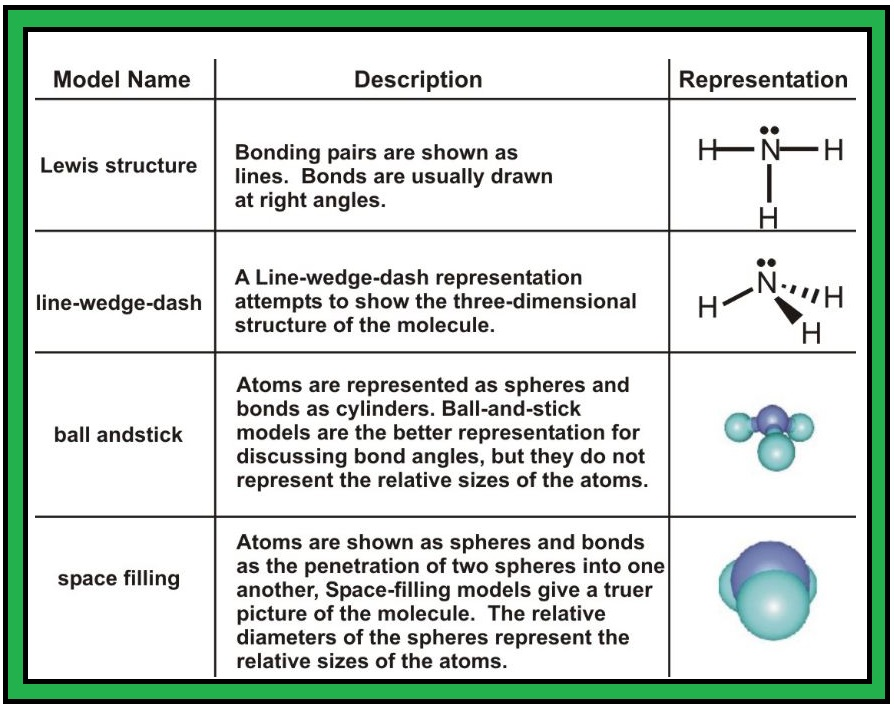
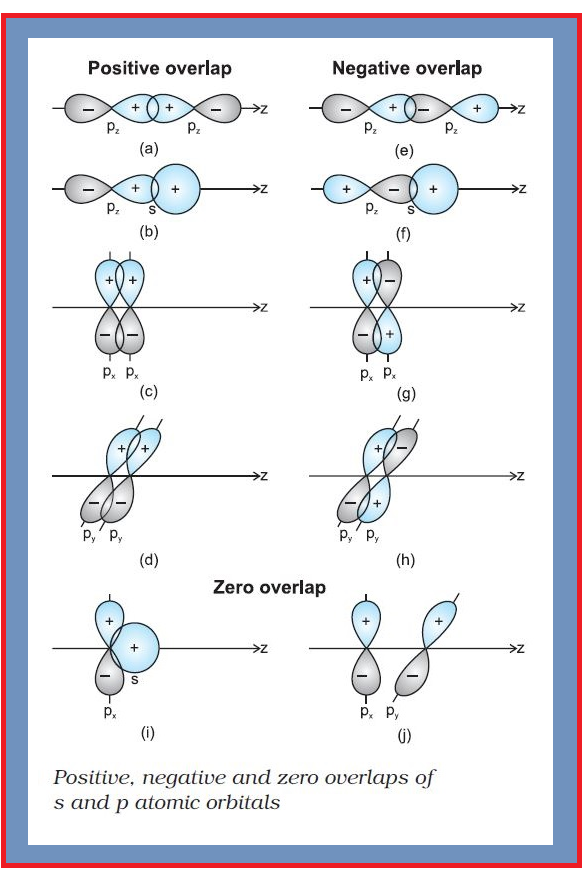
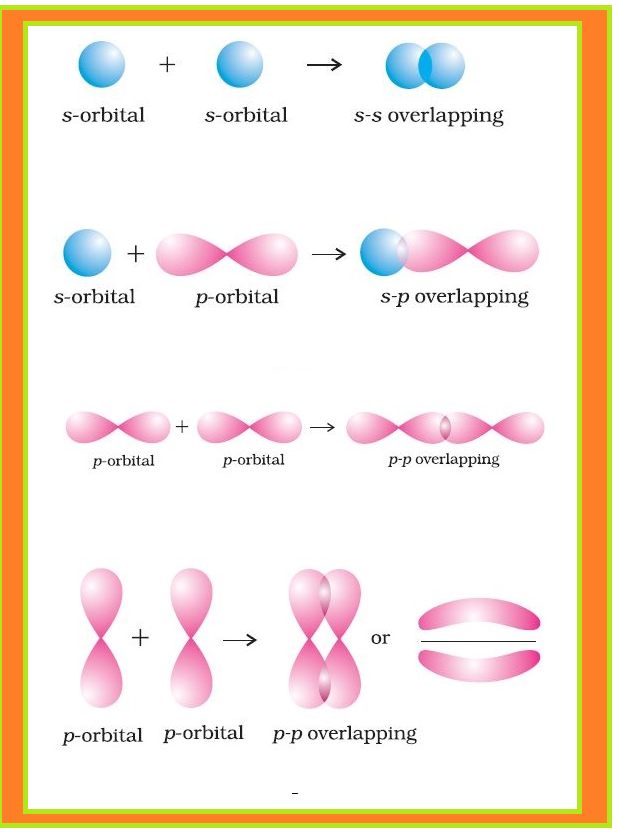
Valence Bond Theory :
`=>` Lewis approach helps in writing the structure of molecules but it fails to explain the formation of chemical bond.
● It also fails to give any reason for the difference in bond dissociation enthalpies and bond lengths in molecules like `H_2 (435.8 kJ mol^(-1), 74 p m)` and `F_2 (150.6 kJ mol^(-1), 42 p m)`, although in both the cases a single covalent bond is formed by the sharing of an electron pair between the respective atoms.
● It also gives no idea about the shapes of polyatomic molecules.
`=>` Similarly the VSEPR theory gives the geometry of simple molecules but theoretically, it does not explain them and also it has limited applications.
`=>` To overcome these limitations the two important theories based on quantum mechanical principles are introduced.
● These are valence bond (VB) theory and molecular orbital (MO) theory.
`=>` Valence bond theory was introduced by Heitler and London (1927) and developed further by Pauling and others.
`=>` Valence bond theory is based on the knowledge of atomic orbitals, electronic configurations of elements, the overlap criteria of atomic orbitals, the hybridization of atomic orbitals and the principles of variation and superposition.
● For the sake of convenience, valence bond theory has been discussed in terms of qualitative and non-mathematical treatment only.
`=>` To start with, let us consider the formation of hydrogen molecule which is the simplest of all molecules.
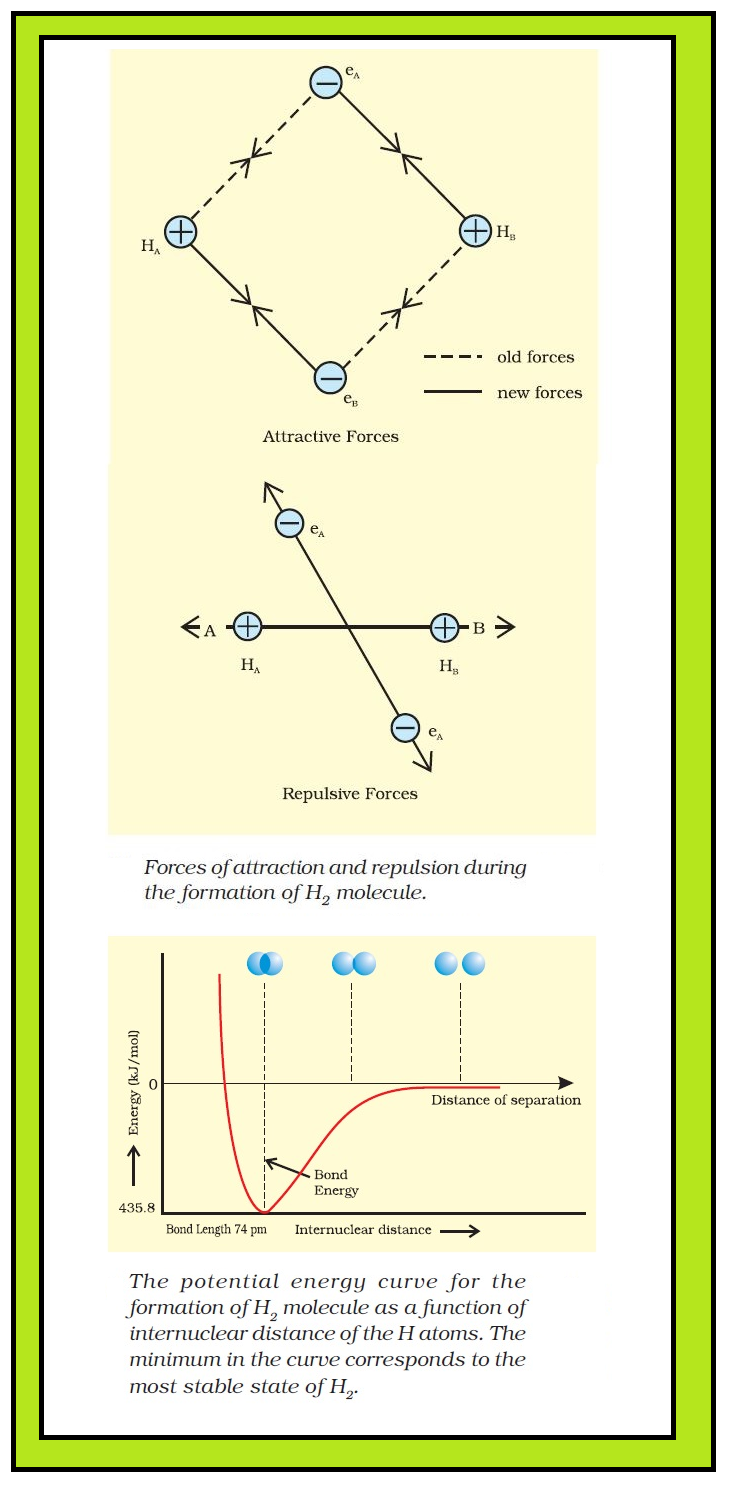
● Consider two hydrogen atoms `A` and `B` approaching each other having nuclei `N_A` and `N_B` and electrons present in them are represented by `e_A` and `e_B`.
● When the two atoms are at large distance from each other, there is no interaction between them.
● As these two atoms approach each other, new attractive and repulsive forces begin to operate.
● `color{green}("Attractive forces arise between")` :
(i) nucleus of one atom and its own electron that is `N_A – e_A` and `N_B– e_B`.
(ii) nucleus of one atom and electron of other atom i.e., `N_A– e_B, N_B– e_A.`
● `color{green}("Similarly repulsive forces arise between")` :
(i) electrons of two atoms like `e_A – e_B,`
(ii) nuclei of two atoms `N_A – N_B.`
● Attractive forces tend to bring the two atoms close to each other whereas repulsive forces tend to push them apart.
● Experimentally, magnitude of new attractive force is more than the new repulsive forces.
● As a result, two atoms approach each other and potential energy decreases and a stage is reached where the net force of attraction balances the force of repulsion and system acquires minimum energy.
● At this stage two hydrogen atoms are said to be bonded together to form a stable molecule having the bond length of `74` pm.
● Since the energy gets released when the bond is formed between two hydrogen atoms, the hydrogen molecule is more stable than that of isolated hydrogen atoms. The energy so released is called as bond enthalpy, which is corresponding to minimum in the curve depicted in Fig.
● Conversely, `435.8` `kJ` of energy is required to dissociate one mole of `H_2` molecule.
`H_2 (g) +435.8 kJ mol^(-1) → H (g) +H (g)`
● It also fails to give any reason for the difference in bond dissociation enthalpies and bond lengths in molecules like `H_2 (435.8 kJ mol^(-1), 74 p m)` and `F_2 (150.6 kJ mol^(-1), 42 p m)`, although in both the cases a single covalent bond is formed by the sharing of an electron pair between the respective atoms.
● It also gives no idea about the shapes of polyatomic molecules.
`=>` Similarly the VSEPR theory gives the geometry of simple molecules but theoretically, it does not explain them and also it has limited applications.
`=>` To overcome these limitations the two important theories based on quantum mechanical principles are introduced.
● These are valence bond (VB) theory and molecular orbital (MO) theory.
`=>` Valence bond theory was introduced by Heitler and London (1927) and developed further by Pauling and others.
`=>` Valence bond theory is based on the knowledge of atomic orbitals, electronic configurations of elements, the overlap criteria of atomic orbitals, the hybridization of atomic orbitals and the principles of variation and superposition.
● For the sake of convenience, valence bond theory has been discussed in terms of qualitative and non-mathematical treatment only.
`=>` To start with, let us consider the formation of hydrogen molecule which is the simplest of all molecules.
● Consider two hydrogen atoms `A` and `B` approaching each other having nuclei `N_A` and `N_B` and electrons present in them are represented by `e_A` and `e_B`.
● When the two atoms are at large distance from each other, there is no interaction between them.
● As these two atoms approach each other, new attractive and repulsive forces begin to operate.
● `color{green}("Attractive forces arise between")` :
(i) nucleus of one atom and its own electron that is `N_A – e_A` and `N_B– e_B`.
(ii) nucleus of one atom and electron of other atom i.e., `N_A– e_B, N_B– e_A.`
● `color{green}("Similarly repulsive forces arise between")` :
(i) electrons of two atoms like `e_A – e_B,`
(ii) nuclei of two atoms `N_A – N_B.`
● Attractive forces tend to bring the two atoms close to each other whereas repulsive forces tend to push them apart.
● Experimentally, magnitude of new attractive force is more than the new repulsive forces.
● As a result, two atoms approach each other and potential energy decreases and a stage is reached where the net force of attraction balances the force of repulsion and system acquires minimum energy.
● At this stage two hydrogen atoms are said to be bonded together to form a stable molecule having the bond length of `74` pm.
● Since the energy gets released when the bond is formed between two hydrogen atoms, the hydrogen molecule is more stable than that of isolated hydrogen atoms. The energy so released is called as bond enthalpy, which is corresponding to minimum in the curve depicted in Fig.
● Conversely, `435.8` `kJ` of energy is required to dissociate one mole of `H_2` molecule.
`H_2 (g) +435.8 kJ mol^(-1) → H (g) +H (g)`